|
Edible spices
|
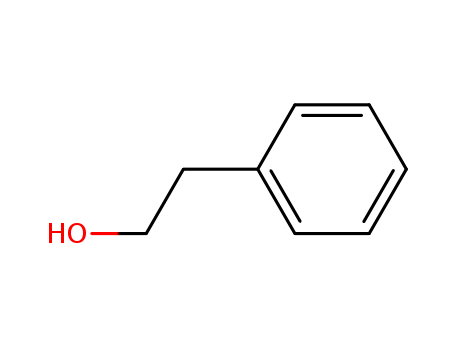
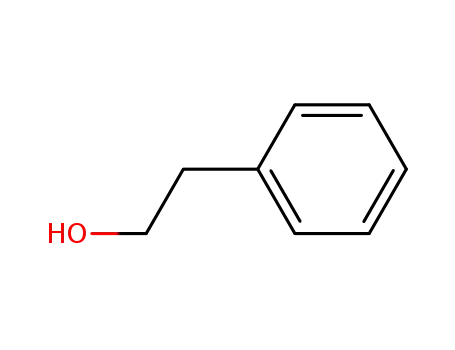
Phenethyl alcohol is a kind of edible spices, and naturally exists in neroli, rose oil, geranium oil and other oils, because it has a soft, pleasant and persistent rose fragrance and is widely used in various kinds of flavors and cigarette flavor. It is dispensing rose scent, food additives, the main raw material for rose scent flavor, stable on alkali, which are widely used in soap fragrance, is essence blending all rose scent series of spices, because it does not dissolve in water, it is often used in the making up water, soap and orange flower, purple, etc. It is also used in the blending of flavor. Because the Phenethyl alcohol has a good antibacterial efficiency, it can be used in the ophthalmic solution. At present there are main three synthesis methods as following:
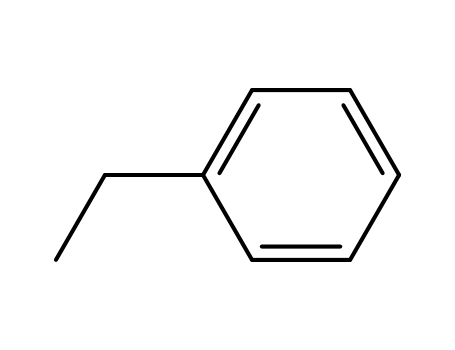
1, by styrene via halogenation, saponification, hydrogenation, distillation.
2, and microorganism fermentation in yeast by bioconversion.
3, calcium carbide, benzene as raw material preparation of benzyl ethanol, reaction equations are as follows:
1)CaC2+2H2O=Ca(OH)2+C2H2
2)C6H6+C2H2=C6H6CHCH2(Styrene)
3)C6H6CHCH2+H2O=C6H6CH2CH2OH(Phenylethyl alcohol) |
|
Occurrence
|
Reported found (as is or esterified) in several natural products: rose concentrate, rose absolute (60% or more)
and rose distillation waters; also found in the essential oils of neroli, ylang-ylang, narcissus, hyacinth, lily, tea leaves, Michelia champaca,
Pandamus odoratissimus, Congo and Réunion geranium, tobacco and other oils. It has been identified in wines. It has also been reported found in over 200 foods and beverages including apple, apricot, orange juice, orange peel, many berries, bilberry, cherry,
grapefruit, peach, raisin, blackberry, guava, grapes, melon, papaya, asparagus, cabbage, leek, potato, rutabaga, tomato, Mentha
oils, cinnamon, ginger, breads, butter, saffron, mustard, mango, many cheeses, butter, milk, cooked chicken, cognac, hop oil, beer,
rum, whiskies, cider, sherry, cocoa, coffee, tea, nuts, oats, honey, soybean, coconut meat, avocado, olive, passion fruit, plum, beans,
mushroom, starfruit, mango, tamarind, fruit brandies, fig, gin, rice, quince, radish, litchi, sukiyaki, calamus, licorice, buckwheat,
watercress, elderberry fruit, kiwifruit, loquat, Tahiti and Bourbon vanilla, mountain papaya, endive, lemon balm, clary sage, shrimps,
crab, Chinese quince, lamb’s lettuce, truffle and maté. |