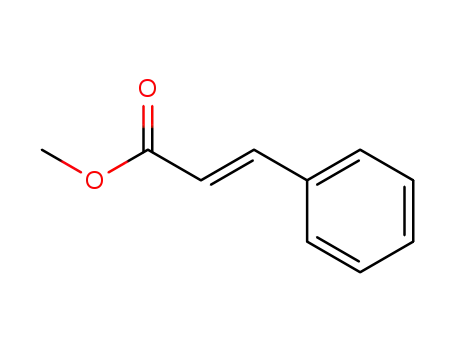
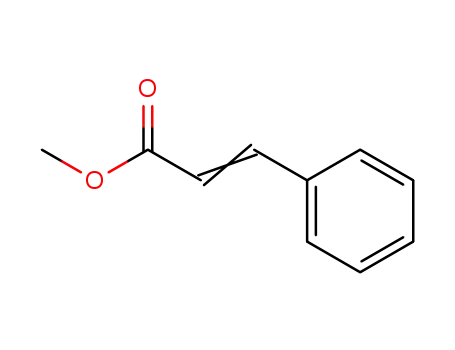
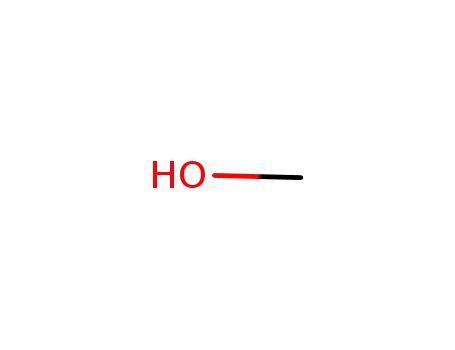
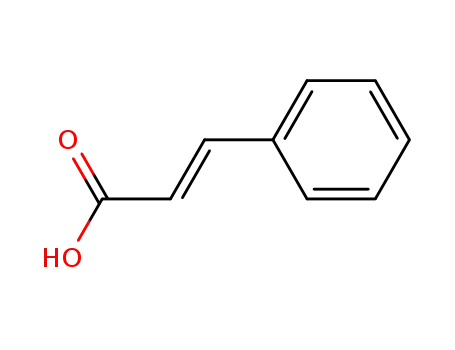
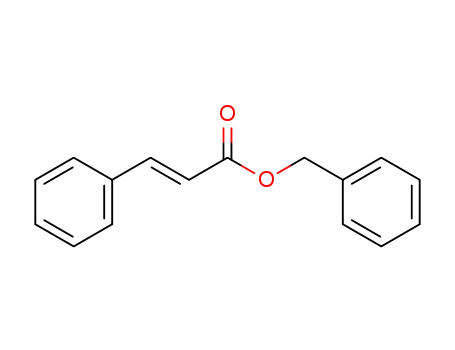
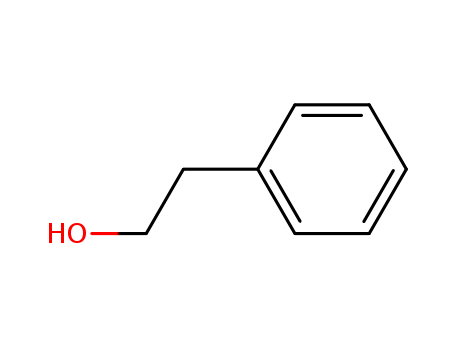
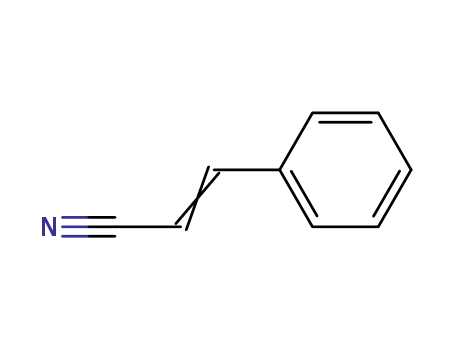Palladium nanoparticles on β-cyclodextrin functionalised graphene nanosheets: A supramolecular based heterogeneous catalyst for C-C coupling reactions under green reaction conditions
Putta, Chandrababu,Sharavath, Vittal,Sarkar, Suprabhat,Ghosh, Sutapa
, p. 6652 - 6660 (2015)
The use of functional properties of nati...
2-Pyridyl substituents enhance the activity of palladium-phospha-adamantane catalysts for the methoxycarbonylation of phenylacetylene
Shuttleworth, Timothy A.,Miles-Hobbs, Alexandra M.,Pringle, Paul G.,Sparkes, Hazel A.
, p. 125 - 137 (2017)
The synthesis of a series of CgPAr ligan...
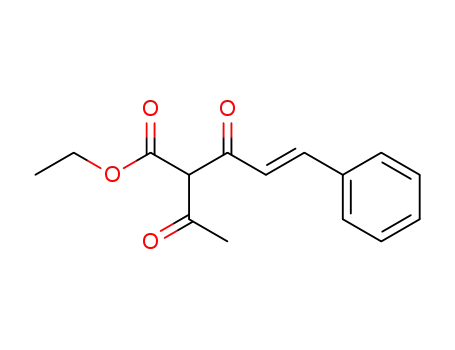
Chalcone and cinnamate synthesis via one-pot enol silane formation-Mukaiyama aldol reactions of ketones and acetate esters
Downey, C. Wade,Glist, Hadleigh M.,Takashima, Anna,Bottum, Samuel R.,Dixon, Grant J.
, p. 3080 - 3083 (2018)
Aryl alkyl ketones, acetate esters, and ...
Highly reactive heterogeneous Heck and hydrogenation catalysts constructed through 'bottom-up' nanoparticle self-assembly
Galow, Trent H.,Drechsler, Ulf,Hanson, Jarrod A.,Rotello, Vincent M.
, p. 1076 - 1077 (2002)
Polymer-mediated self-assembly of functi...
Simultaneous immobilization of a matrix containing palladium and phase transfer catalyst on silica nanoparticles: Application as a recoverable catalyst for the Heck reaction in neat water
Hajipour, Abdol R.,Azizi, Ghobad
, p. 20704 - 20708 (2014)
Simultaneous covalent anchoring of a pho...
Palladium(II)-catalyzed phenylation of unsaturated compounds using phenylantimony chlorides under air
Matoba, Kazutaka,Motofusa, Shin-Ichi,Sik Cho, Chan,Ohe, Kouichi,Uemura, Sakae
, p. 3 - 10 (1999)
Diphenylantimony chloride and phenylanti...
Highly active Pd(II) cyclometallated imine catalysts for the Heck reaction
Ohff, Manuela,Ohff, Andreas,Milstein, David
, p. 357 - 358 (1999)
The new cyclopalladated, phosphine-free ...
Silica hybrid material containing Pd-NHC complex as heterogeneous catalyst for Mizoroki-Heck reactions
Polshettiwar, Vivek,Hesemann, Peter,Moreau, Jo?l J.E.
, p. 5363 - 5366 (2007)
A silica hybrid material containing pall...
Magnetic dendritic polymer nanocomposites as supports for palladium: A highly efficient and reusable catalyst for Mizoroki-Heck and Suzuki-Miyaura coupling reactions
Ma, Rong,Yang, Pengbo,Bian, Fengling
, p. 4748 - 4756 (2018)
A novel catalyst Fe3O4@SiO2-Dendrimer-Pd...
Synthesis of novel palladacycles and their application in heck and Suzuki reactions under aerobic conditions
Xiong, Zhengchang,Wang, Nengdong,Dai, Mingji,Li, Ang,Chen, Jiahua,Yang, Zhen
, p. 3337 - 3340 (2004)
(Chemical Equation Presented) Design and...
Sulfilimine palladacycles: Stable and efficient catalysts for carbon-carbon coupling reactions
Thakur, Vinay V.,Ramesh Kumar,Sudalai
, p. 2915 - 2918 (2004)
A new family of sulfilimine-based pallad...
Pd nanosized particles supported on chitosan and 6-deoxy-6-amino chitosan as recyclable catalysts for Suzuki-Miyaura and Heck cross-coupling reactions
Makhubela, Banothile C.E.,Jardine, Anwar,Smith, Gregory S.
, p. 231 - 241 (2011)
Several chitosan and 6-deoxy-6-amino chi...
Cellulose supported Pd(II) complex catalyzed carbon-carbon bonds formation
Sarkar, Shaheen M.,Rashid,Karim, Kaykobad Md. Rezaul,Mustapha, Siti Noor Hidayah,Lian, Yuen Mei,Zamri, Normaiza,Khan, Md. Maksudur Rahman,O'Reilly, Emmet J.,Rahman, Md. Lutfor
, p. 2856 - 2861 (2019)
Corn-cobs are an agro-industrial waste a...
Preparation of microcapsule-supported palladium catalyst using SPG (Shirasu Porous Glass) emulsification technique
Liu, Ying,Feng, Xiu Juan,Bao, De Cai,Li, Kai Xiao,Bao, Ming
, p. 979 - 982 (2010)
A new method for the preparation of micr...
Catalysis of the Heck-type reaction of alkenes with arylboronic acids by silica-supported rhodium: An efficient phosphine-free reusable catalytic protocol
Trivedi, Rajiv,Roy, Sarabindu,Roy, Moumita,Sreedhar,Kantam, M. Lakshmi
, p. 1575 - 1578 (2007)
A 3-aminopropyl-functionalized silica-su...
Direct Oxidation of Acetals and Aldehydes to Esters
Takeda, Takanobu,Watanabe, Hidenori,Kitahara, Takeshi
, p. 1149 - 1150 (1997)
One step conversion of acetals and aldeh...
A convenient synthesis of 2-(6-methoxy-2-naphthyl)propenoic acid (a naproxen precursor)
Scrivanti,Matteoli
, p. 9015 - 9018 (1995)
The title compound has been synthesised ...
Pd–Schiff base complex supported on Fe3O4 magnetic nanoparticles: A new and highly efficient reusable catalyst for C–C bond formation in water
Lei, Lei
, (2019)
A protocol is introduced for the prepara...
PALLADIUM-CATALYZED OXIDATIVE COUPLING OF AROMATIC COMPOUNDS WITH OLEFINS USING t-BUTYL PERBENZOATE AS A HYDROGEN ACCEPTER
Tsuji, Jiro,Nagashima, Hideo
, p. 2699 - 2702 (1984)
Benzene and furans undergo oxidative cou...
Palladium-catalyzed Mizoroki-Heck type reaction with aryliodine diacetates using hydrazone ligand
Mino, Takashi,Watanabe, Kohei,Abe, Taichi,Kogure, Taketo,Sakamoto, Masami
, p. 2015 - 2021 (2013)
We developed a palladium-catalyzed Mizor...
Palladium-catalyzed Mizoroki-Heck-type reactions of [Ph2SRfn][OTf] with alkenes at room temperature
Wang, Shi-Meng,Song, Hai-Xia,Wang, Xiao-Yan,Liu, Nan,Qin, Hua-Li,Zhang, Cheng-Pan
, p. 11893 - 11896 (2016)
The first Pd-catalyzed Mizoroki-Heck-typ...
Crystalline hybrid solid materials of palladium and decamethylcucurbit[5] uril as recoverable precatalysts for heck cross-coupling reactions
Li, Hongfang,Lue, Jian,Lin, Jingxiang,Huang, Yuanbiao,Cao, Minna,Cao, Rong
, p. 15661 - 15668 (2013)
A series of M-Pd-Me10CB[5] (M=Li, Na, K,...
A click strategy for the immobilization of palladium nanoparticles onto silica: Efficient and recyclable catalysts for carbon-carbon bond formation under mild reaction conditions
Hajipour, Abdol R.,Abolfathi, Parisa,Mohammadsaleh, Fatemeh
, p. 78080 - 78089 (2016)
An interesting silica-supported nano-pal...
A NOVEL ROUTE TO α,β-UNSATURATED ESTERS VIA A REFORMATSKY-TYPE REACTION USING SODIUM TELLURIDE
Suzuki, Hitomi,Inouye, Masahiko
, p. 403 - 406 (1986)
Sodium telluride, prepared by heating te...
In-situ-generated palladium nanoparticles in novel ionic liquid: an efficient catalytic system for Heck–Matsuda coupling
Gaikwad,Undale,Patil,Pore,Korade,Kamble
, p. 4445 - 4458 (2017)
Abstract: A green, convenient, ecologica...
Chemoselective and stereoselective debromination of vicinal-dibromides with sodium dithionite
Khurana, Jitender M.,Sehgal, Arti
, p. 3791 - 3798 (1996)
A simple and efficient procedure for the...
Sodium 2-(2-pyridin-3-ylethylamino)sulfonate: an efficient ligand and base for palladium-catalyzed Heck reaction in aqueous media
Pawar, Shivaji S.,Dekhane, Deepak V.,Shingare, Murlidhar S.,Thore, Shivaji N.
, p. 4252 - 4255 (2008)
The first successful Pd(OAc)2, N-donor l...
-
Spencer,Thorpe
, p. C8 (1975)
-
A dithizone-functionalized polystyrene resin-supported Pd(II) complex as an effective catalyst for Suzuki, Heck, and copper-free Sonogashira reactions under aerobic conditions in water
Bakherad, Mohammad,Jajarmi, Saeideh
, p. 152 - 159 (2013)
A novel polystyrene-supported palladium(...
Cobalt supported on dendronized magnetic nanoparticles: A new highly efficient and recyclable catalyst for the Mizoroki–Heck cross-coupling reaction
Arghan, Maryam,Koukabi, Nadiya,Kolvari, Eskandar
, (2019)
Polyamidoamine (PAMAM) is one of the mos...
Methoxylation of Acyl Fluorides with Tris(2,4,6-trimethoxyphenyl)phosphine via C-OMe Bond Cleavage under Metal-Free Conditions
Ishida, Takumi,Nishihara, Yasushi,Wang, Xiu,Wang, Zhenhua
, p. 7526 - 7533 (2020)
Acyl fluorides are subjected to methoxyl...
Silica-Supported Ni(II)–DABCO Complex: An Efficient and Reusable Catalyst for the Heck Reaction
Hajipour, Abdol R.,Abolfathi, Parisa
, p. 188 - 195 (2017)
Abstract: An interesting nickel-based ca...
Fluorapatite-supported palladium catalyst for Suzuki and Heck coupling reactions of haloarenes
Kantam, M. Lakshmi,Kumar, K. B. Shiva,Srinivas,Sreedhar
, p. 1141 - 1149 (2007)
A fluorapatite-supported palladium catal...
DIRECT SYNTHESIS OF Z-UNSATURATED ESTERS. A USEFUL MODIFICATION OF THE HORNER-EMMONS OLEFINATION.
Still, W. Clark,Gennari, Cesare
, p. 4405 - 4408 (1983)
New phosphonoester reagents and reaction...
Pd complex of an NNN pincer ligand supported on γ-Fe2O3@SiO2 magnetic nanoparticles: A new catalyst for Heck, Suzuki and Sonogashira coupling reactions
Sobhani, Sara,Zeraatkar, Zohre,Zarifi, Farzaneh
, p. 7076 - 7085 (2015)
In this study, a Pd complex of bis(imino...
Palladium-catalysed Vinylation of Organic Halides under Solid-Liquid Phase Transfer Conditions
Jeffery, Tuyet
, p. 1287 - 1289 (1984)
Heck-type reactions involving organic ha...
Arylation and Alkylation of Olefins by Arylamines or Hydrazines via Carbon-Nitrogen Bond Cleavage in the Presence of Palladium(II) Salts
Akiyama, Fumitaka,Miyazaki, Hiroshi,Kaneda, Kiyotomi,Teranishi, Shiichiro,Fujiwara, Yuzo,et al.
, p. 2359 - 2361 (1980)
Arylamines (ArNH2) have been found to fu...
Salen-porphyrin-based conjugated microporous polymer supported Pd nanoparticles: Highly efficient heterogeneous catalysts for aqueous C-C coupling reactions
Ju, Pengyao,Wu, Shujie,Su, Qing,Li, Xiaodong,Liu, Ziqian,Li, Guanghua,Wu, Qiaolin
, p. 2660 - 2666 (2019)
The salen-porphyrin based conjugated mic...
Palladium-catalysed cross-coupling reactions in supercritical carbon dioxide
Early,Gordon,Carroll,Holmes,Shute,McConvey
, p. 1966 - 1967 (2001)
Heck and Suzuki reactions proceed in goo...
Amidoxime modified PAN supported palladium complex: A greener and efficient heterogeneous catalyst for heck reaction
Sruthi, Pambingal Rajan,Sarika, Vijayalekshmi,Suku, Arya,Krishnan, Aravind,Anas, Saithalavi
, (2020)
Development of a facile and greener appr...
Manganese phthalocyanine immobilized on silica gel: Efficient and recyclable catalyst for single-step oxidative esterification of aldehydes with alcohols
Sharma,Gulati, Shikha
, p. 291 - 303 (2012)
The functionalization of silica gel was ...
Pd Nanoparticles Stabilized on the Cross-Linked Melamine-Based SBA-15 as a Catalyst for the Mizoroki–Heck Reaction
Amiri, Mandana,Bezaatpour, Abolfazl,Mikkola, Jyri-Pekka,Murzin, Dmitry Yu.,Nuri, Ayat,Vucetic, Nemanja
, (2021)
Mesoporous SBA-15 silicate with a high s...
Poly(hydroxamic acid) palladium catalyst for heck reactions and its application in the synthesis of Ozagrel
Sarkar, Shaheen M.,Rahman, Md. Lutfor,Chong, Kwok Feng,Yusoff, Mashitah Mohd
, p. 103 - 110 (2017)
Bio-waste corn-cob cellulose supported p...
Homoleptic cis- and trans-palladium(II) bis(guanidinato) complexes derived from N-aryl-N′,N″-di(pyridin-2-yl)- and N-aryl-N′,N″-bis(6-methylpyridin-2-yl)guanidines: Catalysts for Heck-Mizoroki coupling reactions
Mishra, Vishwesh,Thomas, Jisha Mary,Chinnappan, Sivasankar,Thirupathi, Natesan
, p. 1 - 17 (2019)
N-Aryl-N′,N″-di(pyridin-2-yl)- and N-ary...
Thiophene Methanimine–Palladium Schiff Base Complex Anchored on Magnetic Nanoparticles: A Novel, Highly Efficient and Recoverable Nanocatalyst for Cross-Coupling Reactions in Mild and Aqueous Media: Γ-Fe2O3/AEPH2-TC-Pd Catalyzed Suzuki–Miyaura and Heck–Mizoroki Reactions
Jahanshahi, Roya,Akhlaghinia, Batool
, p. 2640 - 2655 (2017)
Abstract: In this study, a novel thiophe...
A heterometal (Pd–Pb) organic framework: synthesis, structure and heterogeneous catalytic application
Ren, Yanwei,Jiang, Ou,Li, Jiawei,Zeng, Hang
, p. 699 - 704 (2016)
A heterometallic organic framework {Pb[P...
Aminophosphine Palladium(0) Complex Supported on ZrO2 Nanoparticles (ZrO2@AEPH2-PPh2-Pd(0)) as an Efficient Heterogeneous Catalyst for Suzuki–Miyaura and Heck–Mizoroki Reactions in Green Media
Razavi, Nasrin,Akhlaghinia, Batool,Jahanshahi, Roya
, p. 360 - 373 (2017)
Abstract: A new aminophosphine palladium...
Piperazine-derived palladium complexes immobilised on a Merrifield resin as a catalyst for the Heck reaction
Wu, Xiang-Mei,Pei, Wen
, p. 6 - 8 (2008)
A palladium catalyst supported on a Merr...
Synthesis and crystal structures of a series of (μ-thiophenolato)(μ-pyrazolato-N,N′) double bridged dipalladium(II) complexes and their application in Mizoroki-Heck reaction as highly efficient catalysts
Khadir, Narjes,Tavakoli, Ghazal,Assoud, Abdeljalil,Bagherzadeh, Mojtaba,Boghaei, Davar M.
, p. 107 - 117 (2016)
Three new binucleating S-protected ligan...
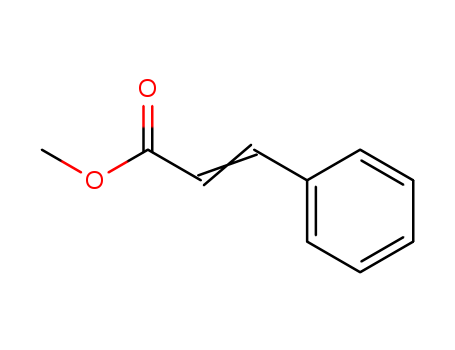
Novel diphosphine-modified palladium catalysts for oxidative carbonylation of styrene to methyl cinnamate
Bianchini, Claudio,Mantovani, Giuseppe,Meli, Andrea,Oberhauser, Werner,Brueggeller, Peter,Stampfl, Thomas
, p. 690 - 698 (2001)
The oxidative carbonylation of styrene h...
Synthesis of α-diazoesters from α-hydrazonoesters: Utilization of α-hydrazonoesters and α-diazoesters for convenient interconversion
Yasui, Eiko,Ishimine, Kanako,Nitanai, Sohta,Hatakeyama, Nanami,Nagumo, Shinji
, (2022/04/07)
We have developed a novel method to synt...
Click amidations, esterifications and one–pot reactions catalyzed by Cu salts and multimetal–organic frameworks (M–MOFs)
Armentano, Donatella,Ferrando-Soria, Jesús,Greco, Rossella,Leyva-Pérez, Antonio,Palomar-De Lucas, Brenda,Pardo, Emilio,Tiburcio, Estefanía
, (2022/03/17)
Amides and esters are prevalent chemical...
A Straightforward, Purification-Free Procedure for the Synthesis of Ando and Still-Gennari Type Phosphonates
Janicki, Ignacy,Kie?basiński, Piotr
, p. 378 - 382 (2021/10/21)
Z-Selective Still-Gennari and Ando modif...
Visible-Light-Driven Isocyanide Insertion to o-Alkenylanilines: A Route to Isoindolinone Synthesis
Dahiya, Anjali,Das, Bubul,Sahoo, Ashish Kumar,Patel, Bhisma K.
, p. 966 - 973 (2022/02/07)
A visible-light-mediated intermolecular ...