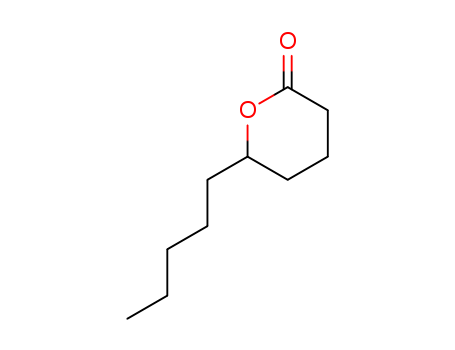
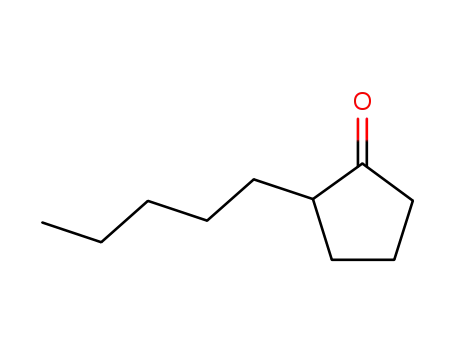
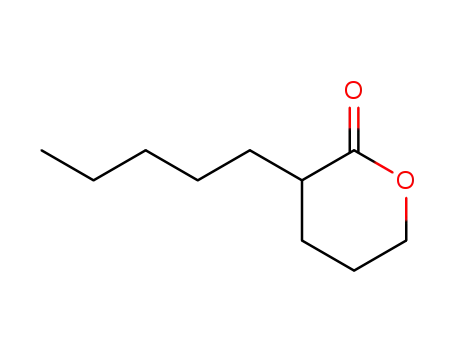
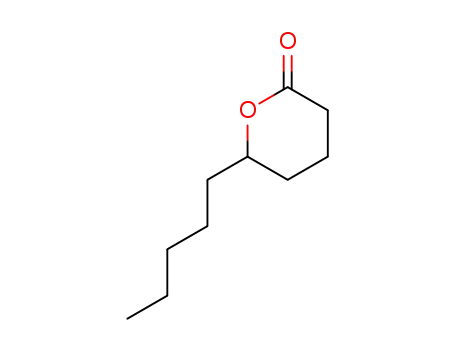
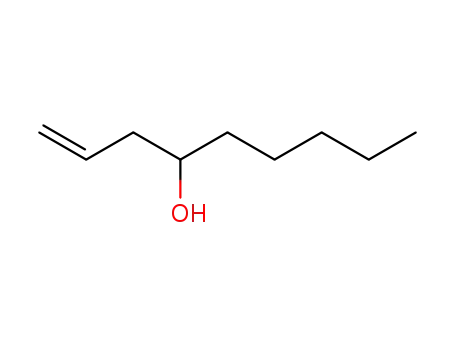
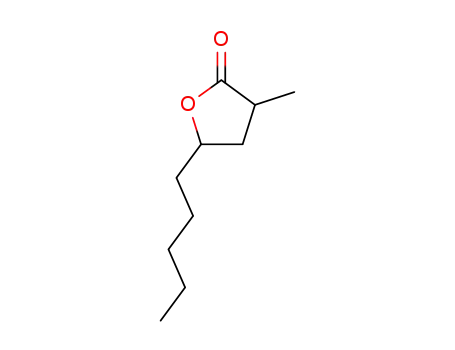
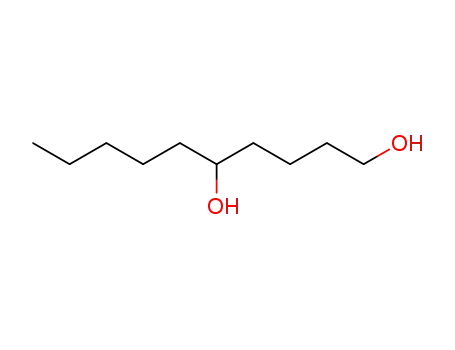
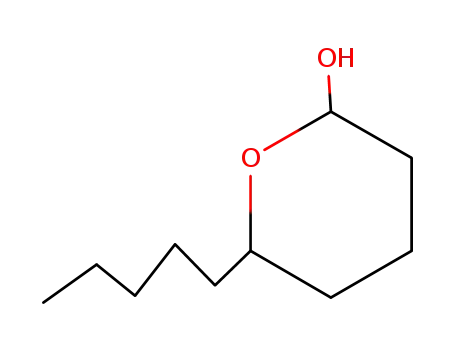
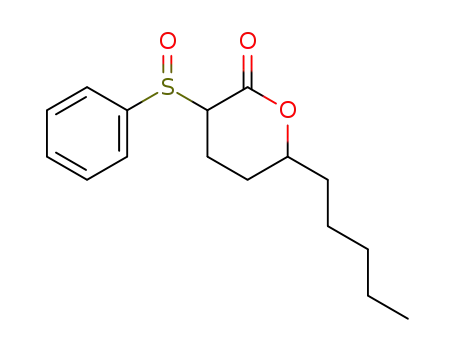
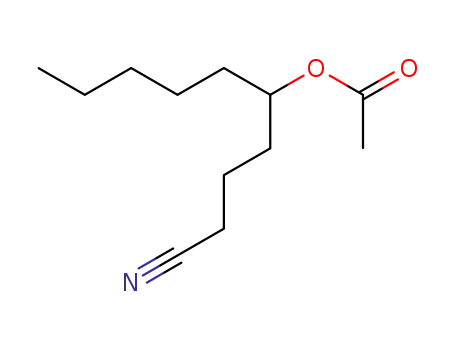
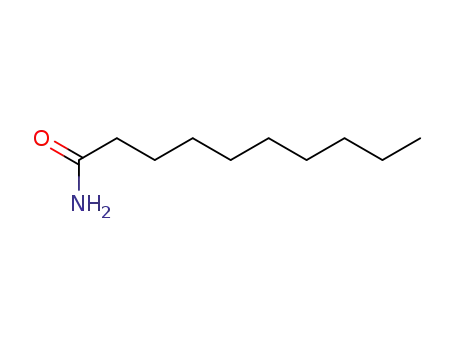
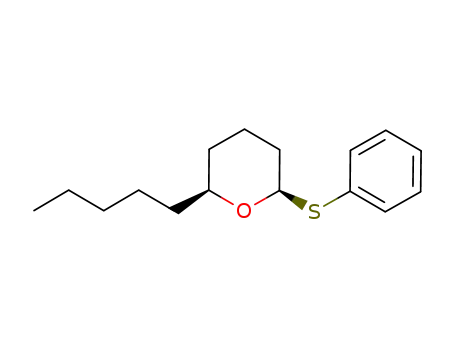
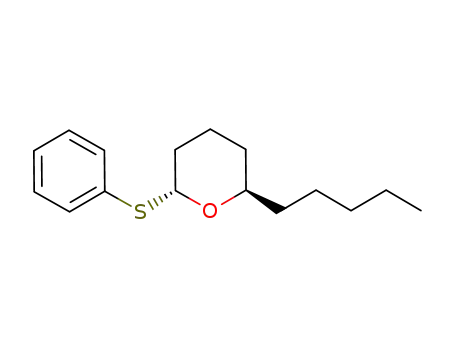
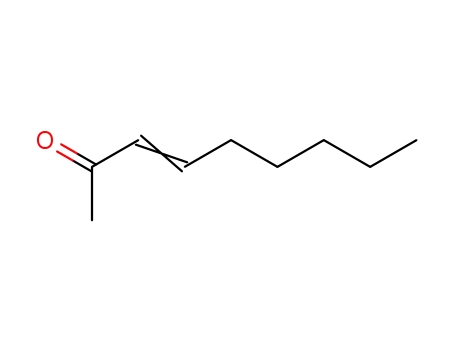
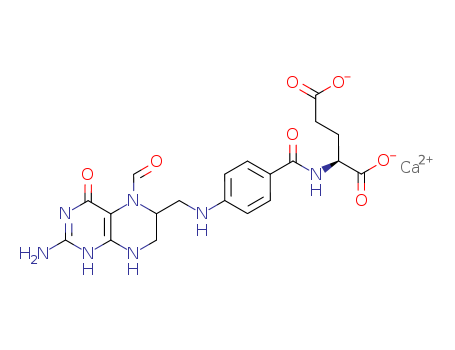
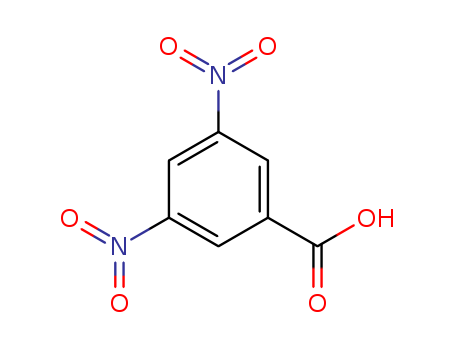
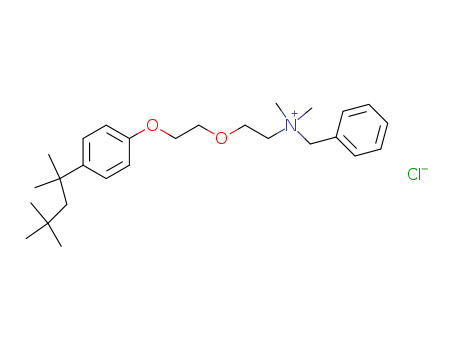
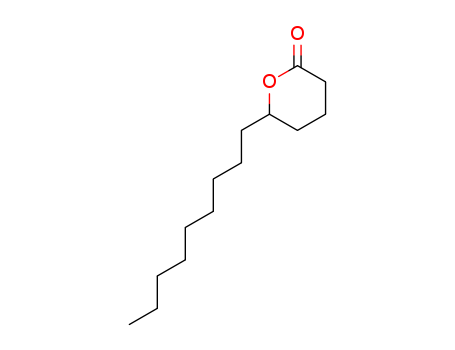
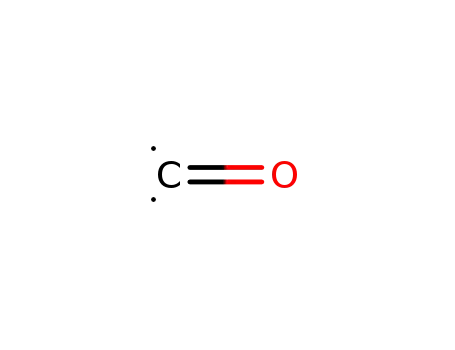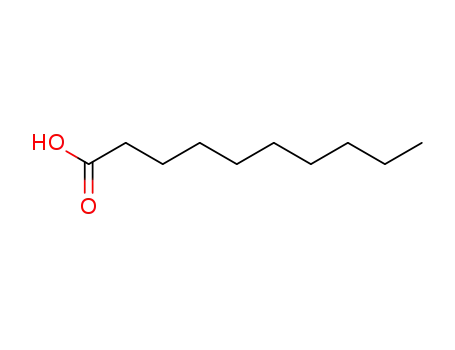| Conditions |
Yield |
|
With
dihydrogen peroxide;
scandium(III) tris[bis(perfluorooctanesulfonyl)imide];
In
various solvent(s);
at 20 ℃;
for 5h;
Title compound not separated from byproducts.;
|
|
|
With
dihydrogen peroxide;
lithium hydroxide;
In
Ethyl propionate;
at 100 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium hydroxide;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
sodium tert-pentoxide;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium hydroxide;
In
tert-Amyl alcohol; water;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
sodium hydrogencarbonate;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
sodium hydrogencarbonate;
In
tert-Amyl alcohol; water;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
magnesium 2-ethylhexanoate;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
sodium hydroxide;
In
tert-Amyl alcohol; water;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium bromide;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
potassium acetate;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
sodium 2,2,2-trifluoroacetate;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
sodium 2-ethylhexanoic acid;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium acetate;
In
water; chlorobenzene;
at 110 ℃;
for 6h;
Reactivity;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium acetate;
In
1,4-dioxane; water;
at 110 ℃;
for 6h;
Reactivity;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium acetate;
In
Isopropyl acetate; water;
at 110 ℃;
for 6h;
Reactivity;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium acetate;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium acetate;
In
tert-Amyl methyl ether; water;
at 110 ℃;
for 6h;
Reactivity;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
calcium carbonate;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
sodium peroxide;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
Na(polyacrylate);
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium pentane-2,4-dionate;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
lithium carbonate;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|
|
With
dihydrogen peroxide;
barium carbonate;
In
water; Ethyl propionate;
at 110 ℃;
for 6h;
Heating / reflux;
|
|