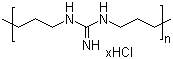
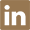|
Chemical Properties
|
Clear colorless oil |
|
Uses
|
Mineral oil is a lubricant and is used as
a solvent for inks in the printing industry. |
|
Production Methods
|
Mineral oil is obtained by distillation of petroleum. The lighter
hydrocarbons are first removed by distillation and the residue is
then redistilled between 330–390°C. The distillate is chilled and the
solid fractions are removed by filtration. The filtrate is then further
purified and decolorized by high-pressure hydrogenation or sulfuric
acid treatment; the purified filtrate is then filtered through
adsorbents. The liquid portion obtained is distilled and the portion
boiling below 360°C is discarded. A suitable stabilizer may be
added to the mineral oil; |
|
Brand name
|
Balneol
(Solvay Pharmaceuticals); Neo-Cultol (Fisons); Nujol
(Schering-Plough HealthCare). |
|
General Description
|
Transparent colorless oily liquid. Practically tasteless and odorless, even when warmed. |
|
Air & Water Reactions
|
Flammable. Insoluble in water. |
|
Reactivity Profile
|
OIL. [MINERAL] may be incompatible with strong oxidizing agents like nitric acid. Charring may occur followed by ignition of unreacted oil and other nearby combustibles. In other settings, mostly unreactive. Not affected by aqueous solutions of acids, alkalis, most oxidizing agents, and most reducing agents. Burns exothermically when heated sufficiently or when ignited in the presence of air, oxygen or strong oxidizing agents. |
|
Hazard
|
Inhalation of vapor or particulates can causeaspiration pneumonia. Combustible liquid. Upperrespiratory tract irritant. Questionable carcinogen. |
|
Health Hazard
|
Highly refined mineral oil mist
is of low toxicity. |
|
Fire Hazard
|
PARAFFIN is combustible. |
|
Pharmaceutical Applications
|
Mineral oil is used primarily as an excipient in topical pharmaceutical
formulations, where its emollient properties are exploited as an
ingredient in ointment bases. It is additionally used in
oil-in-water emulsions,as a solvent, and as a lubricant in
capsule and tablet formulations, and to a limited extent as a mold-release agent for cocoa butter suppositories. It has also been used in
the preparation of microspheres and as a vaccine adjunct.
Therapeutically, mineral oil has been used as a laxative.It is indigestible and thus has limited absorption.
Mineral oil is used in ophthalmic formulations for its lubricant
properties. It is also used in cosmetics and some food products. |
|
Safety Profile
|
A human teratogen by
inhalation that causes testicular tumors in
the fetus. Inhalation of vapor or particulates
can cause aspiration pneumonia. A skin and
eye irritant. Highly purified food grades are
of low toxicity. Questionable human
carcinogen producing gastrointestinal
tumors. Slightly combustible liquid when
exposed to heat or flame. To fight fire, use
dry chemical, CO2, foam. When heated to
decomposition it emits acrid smoke and
fumes. |
|
Safety
|
Mineral oil is used as an excipient in a wide variety of
pharmaceutical formulations. It is also used in
cosmetics and in some food products.
Therapeutically, mineral oil has been used in the treatment of
constipation, as it acts as a lubricant and stool softener when taken
orally. Daily doses of up to 45mL have been administered orally,
while doses of up to 120mL have been used as an enema. However,
excessive dosage of mineral oil, either orally or rectally, can result in
anal seepage and irritation, and its oral use as a laxative is not
considered desirable.
Chronic oral consumption of mineral oil may impair the appetite
and interfere with the absorption of fat-soluble vitamins. Prolonged
use should be avoided. Mineral oil is absorbed to some extent when
emulsified and can lead to granulomatous reactions. Similar
reactions also occur upon injection of the oil;injection may
also cause vasospasm.
The most serious adverse reaction to mineral oil is lipoid
pneumonia caused by aspiration of the oil.Mineral oil can
enter the bronchial tree without eliciting the cough reflex.With
the reduction in the use of mineral oil in nasal formulations, the
incidence of lipoid pneumonia has been greatly reduced. However,
lipoid pneumonia has also been associated with the use of mineral
oil-containing cosmetics and ophthalmic preparations.It is
recommended that products containing mineral oil not be used in
very young children, the elderly, or persons with debilitating
illnesses.
Given its widespread use in many topical products, mineral oil
has been associated with few instances of allergic reactions.
The WHO has not specified an acceptable daily intake of
mineral oil given the low concentration consumed in foods.
LD50 (mouse, oral): 22 g/kg |
|
storage
|
Mineral oil undergoes oxidation when exposed to heat and light.
Oxidation begins with the formation of peroxides, exhibiting an
‘induction period’. Under ordinary conditions, the induction period
may take months or years. However, once a trace of peroxide is
formed, further oxidation is autocatalytic and proceeds very
rapidly. Oxidation results in the formation of aldehydes and
organic acids, which impart taste and odor. Stabilizers may be added to retard oxidation; butylated hydroxyanisole, butylated
hydroxytoluene, and alpha tocopherol are the most commonly used
antioxidants.
Mineral oil may be sterilized by dry heat.
Mineral oil should be stored in an airtight container, protected
from light, in a cool, dry place. |
|
Purification Methods
|
Treat the oil with fuming H2SO4 (care), then wash it with water and dilute aqueous NaOH, then percolate it through activated silica gel. Paraffin Wax. Melt the wax in the presence of NaOH, wash it with water until all of the base had been removed. The paraffin is allowed to solidify after each wash. Finally, 5g of paraffin is melted by heating it on a water-bath, then shaken for 20-30minutes with 100mL of boiling water and and dry the melt under vacuum. |
|
Incompatibilities
|
Incompatible with strong oxidizing agents. |
|
Regulatory Status
|
GRAS listed. Accepted in the UK for use in certain food
applications. Included in the FDA Inactive Ingredients Database
(dental preparations; IV injections; ophthalmic preparations; oral
capsules and tablets; otic, topical, transdermal, and vaginal
preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal
Ingredients. |
|
EXPOSURE ROUTES
|
inhalation, skin and/or eye contact
|
|
FIRST AID
|
(See procedures)
Eye:Irrigate immediately
Breathing:Respiratory support
|
|
Consumer Uses
|
ECHA has no public registered data indicating whether or in which chemical products the substance might be used. ECHA has no public registered data on the routes by which this substance is most likely to be released to the environment. |