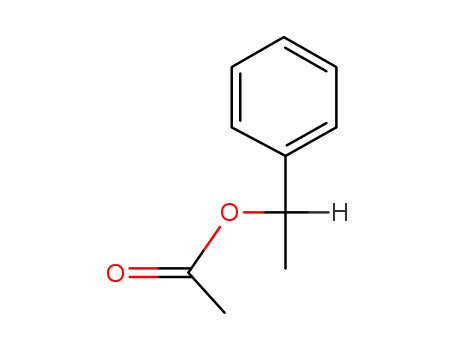
Sale Factory Supply High Purity STYRALLYL ACETATE 93-92-5 Cheapest Price
- Molecular Formula:C10H12O2
- Molecular Weight:164.204
- Appearance/Colour:clear colorless to pale yellow viscous liquid
- Vapor Pressure:0.203mmHg at 25°C
- Melting Point:-60°C
- Refractive Index:n20/D 1.494(lit.)
- Boiling Point:288 °C at 760 mmHg
- Flash Point:170.2 °C
- PSA:26.30000
- Density:1.09 g/cm3
- LogP:2.31070
Styralyl acetate(Cas 93-92-5) Usage
|
Description
|
α-Methylbenzyl acetate has an intensive green odor suggestive of
gardenia with a bitter, acrid taste, interesting on dilution. May be
prepared by acetylation of methyl phenyl carbinol; from benzaldehyde by reacting with magnesium methyl bromide and subsequent acetylation; from 1-bromoethylbenzene and silver acetate in
acetic acid. |
|
Chemical Properties
|
α-Methylbenzyl acetate has an intensive green odor suggestive of gardenia and a bitter, acrid taste, interesting on dilution. |
|
Occurrence
|
Reported found in gardenia flower oil and avocado. |
|
Uses
|
Perfumery, flavoring. |
|
Preparation
|
By acetylation of methyl phenyl carbinol; from benzaldehyde by reacting with magnesium methyl bromide and subse quent acetylation; from 1-bromoethylbenzene and silver acetate in acetic acid. |
|
Taste threshold values
|
Taste characteristics at 12.0 ppm: fruity, berry, green, seedy and slightly nutty. |
|
Synthesis Reference(s)
|
Journal of the American Chemical Society, 88, p. 1833, 1966 DOI: 10.1021/ja00960a057Synthetic Communications, 8, p. 33, 1978 DOI: 10.1080/00397917808062180 |
|
Flammability and Explosibility
|
Nonflammable |
|
Metabolism
|
Although no published data exist on the metabolism of methylphenylcarbinyl acetate, it is likely that hydrolysis to the carbinol and acetic acid is the initial step. Williams (1959) reports studies in which 50% of a dose of (±)-methylphenylcarbinol given to rabbits was excreted as the glucuronide in the urine within 24 hr. There was some evidence of oxidation and demethylation, as mandelic and hippuric acids were found in the urine. Previously, El Masry, Smith |
|
Consumer Uses
|
This substance is used in the following products:
perfumes and fragrances,
air care products,
biocides (e.g. disinfectants, pest control products),
polishes and waxes,
washing & cleaning products and cosmetics and personal care products. Other release to the environment of this substance is likely to occur from:
indoor use as processing aid and outdoor use as processing aid. |
InChI:InChI=1/C10H12O2/c1-8(12-9(2)11)10-6-4-3-5-7-10/h3-8H,1-2H3/t8-/m1/s1
93-92-5 Relevant articles
Noncross-linked polystyrene nanoencapsulation of ferric chloride: A novel and reusable heterogeneous macromolecular Lewis acid catalyst toward selective acetylation of alcohols, phenols, amines, and thiols
Alinejad, Sara,Donyapeyma, Ghazaleh,Rahmatpour, Ali
, (2022/01/24)
Ferric chloride has been successfully na...
Chiral phosphine-phosphoramidite ester ligand as well as preparation method and application thereof
-
Paragraph 0054-0057; 0081-0084; 0095-0096, (2021/05/22)
The invention provides a method for prep...
Sustainable electrochemical decarboxylative acetoxylation of aminoacids in batch and continuous flow
K?ckinger, Manuel,Hanselmann, Paul,Roberge, Dominique M.,Geotti-Bianchini, Piero,Kappe, C. Oliver,Cantillo, David
supporting information, p. 2382 - 2390 (2021/04/12)
Introduction of acetoxy groups to organi...
Method for synthesizing styrallyl acetate from acetophenone
-
Paragraph 0033; 0036; 0063-0129, (2021/03/13)
The invention provides a method for synt...
93-92-5 Process route
-
-
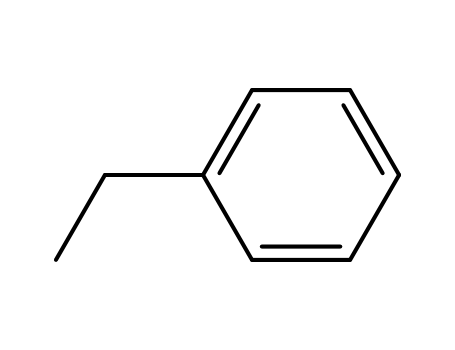
100-41-4,27536-89-6
ethylbenzene
-
-
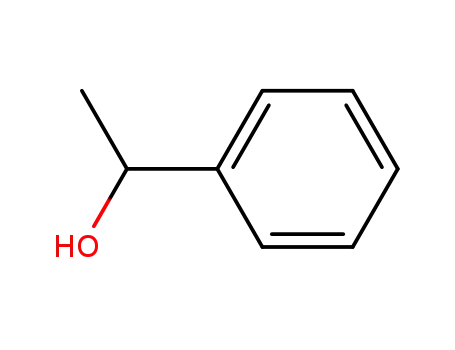
98-85-1,13323-81-4
1-Phenylethanol
-
-
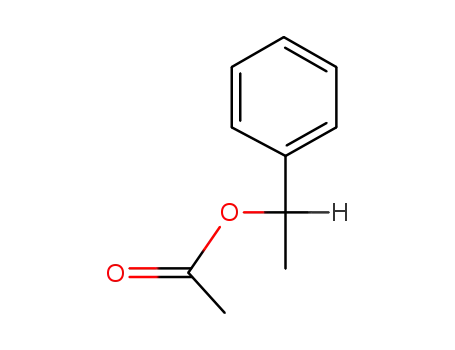
93-92-5,50373-55-2
1-phenylethyl acetate
Conditions
| Conditions |
Yield |
|
With
3,3-dimethyldioxirane;
In
acetone;
for 48h;
Ambient temperature;
|
50%
14%
36 % Chromat.
|
|
With
oxygen; 3,3-dimethyldioxirane;
In
acetone;
for 48h;
under 760 Torr;
Ambient temperature;
|
25%
75 % Chromat.
|
|
With
ammonium peroxydisulfate; copper diacetate;
In
water; acetonitrile;
at 100 ℃;
Yield given. Yields of byproduct given;
|
|
|
With
ammonium peroxydisulfate; copper diacetate;
In
water; acetic acid;
at 100 ℃;
Yield given. Yields of byproduct given;
|
|
|
With
[FeIV(1,1-di(pyridin-2-yl)-N,N-bis(quinolin-2-ylmethyl)methanamine)(O)]2+;
In
acetonitrile;
at 25 ℃;
Inert atmosphere;
Glovebox;
|
34 %Chromat.
26 %Chromat.
5 %Chromat.
|
-
-
4013-34-7,111767-38-5
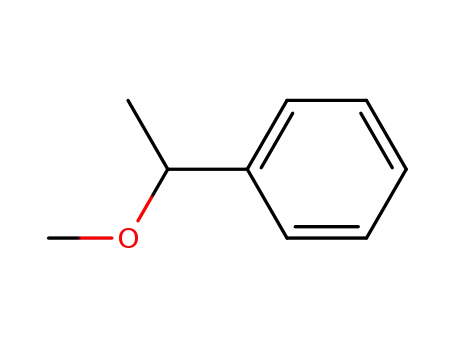
3-phenyl-2-oxabutane
-
-
98-85-1,13323-81-4
1-Phenylethanol
-
-
93-92-5,50373-55-2
1-phenylethyl acetate
Conditions
| Conditions |
Yield |
|
With
ammonium peroxydisulfate; copper diacetate;
In
water; acetonitrile;
at 100 ℃;
Yield given. Yields of byproduct given;
|
|
93-92-5 Upstream products
-
93-59-4
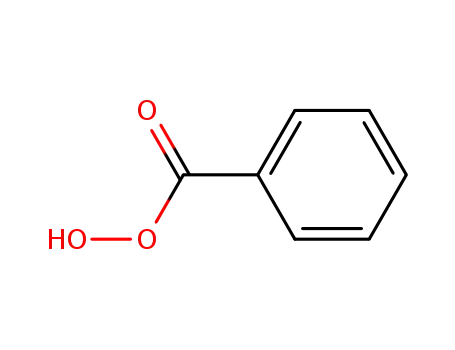
Perbenzoic acid
-
769-59-5
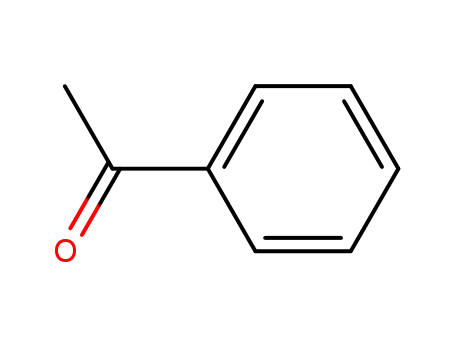
3-phenyl-butan-2-one
-
100-41-4
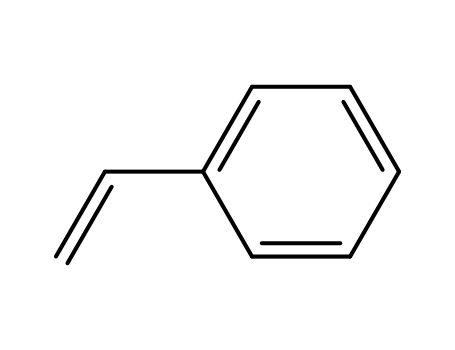
ethylbenzene
-
98-85-1
1-Phenylethanol
93-92-5 Downstream products