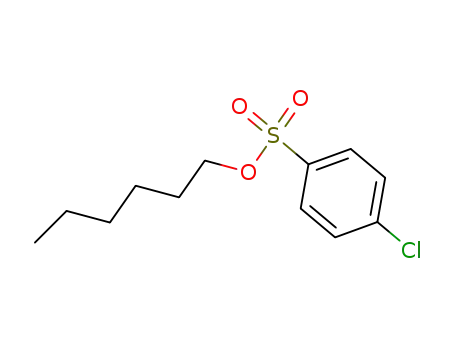
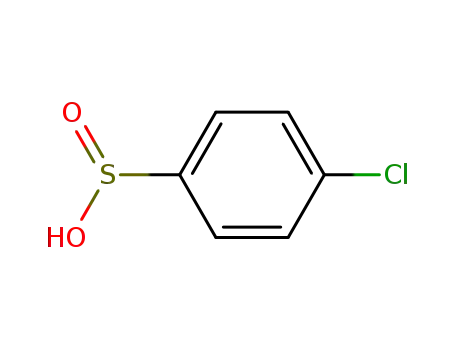
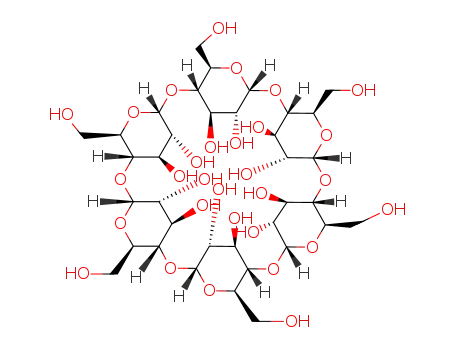
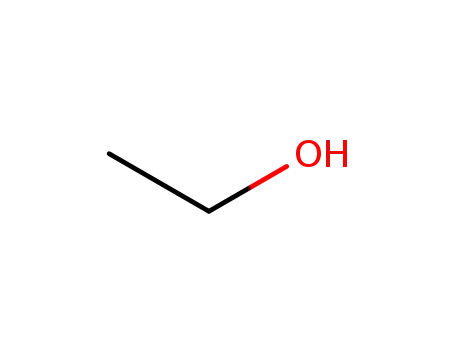
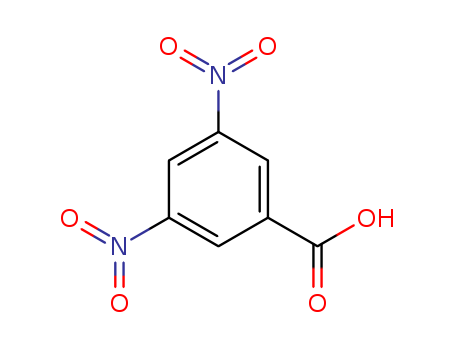
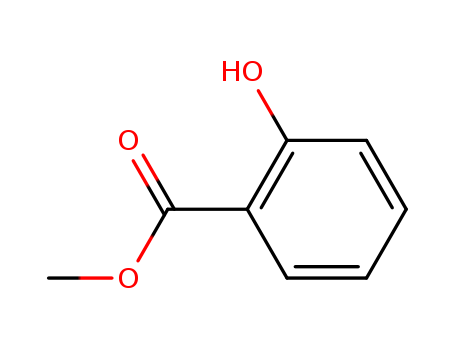
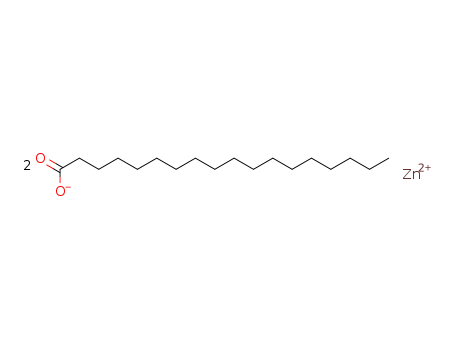Oxyfunctionalization of Alkanes with H2O2 catalysed by Vanadium Silicates
Rao, P. R. Hari Prasad,Ramaswamy, A. V.
, p. 1245 - 1246 (1992)
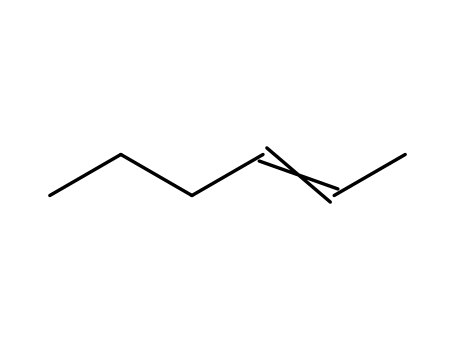
Vanadium silicate molecular sieves can c...
Highly Efficient Photocatalytic Degradation of Dyes by a Copper–Triazolate Metal–Organic Framework
Liu, Chen-Xia,Zhang, Wen-Hua,Wang, Nan,Guo, Penghu,Muhler, Martin,Wang, Yuemin,Lin, Shiru,Chen, Zhongfang,Yang, Guang
, p. 16804 - 16813 (2018)
A copper(I) 3,5-diphenyltriazolate metal...
Interfacial Sites in Ag Supported Layered Double Oxide for Dehydrogenation Coupling of Ethanol to n-Butanol
Zhang, Jian,Shi, Kai,Zhu, Yanru,An, Zhe,Wang, Wanning,Ma, Xiaodan,Shu, Xin,Song, Hongyan,Xiang, Xu,He, Jing
, p. 1095 - 1103 (2021)
Upgrading of ethanol to n-butanol throug...
New selectivities from old catalysts. Occlusion of Grubbs' catalysts in PDMS to change their reactions
Brett Runge,Mwangi, Martin T.,Bowden, Ned B.
, p. 5278 - 5288 (2006)
This article describes new selectivities...
Thermal desulfurization of (Alkoxymethyl)thiiranes
Nalet'Ko,Pervova,Gorbunova,Zapevalov, A. Ya,Toporova,Saloutin
, p. 2120 - 2124 (2014)
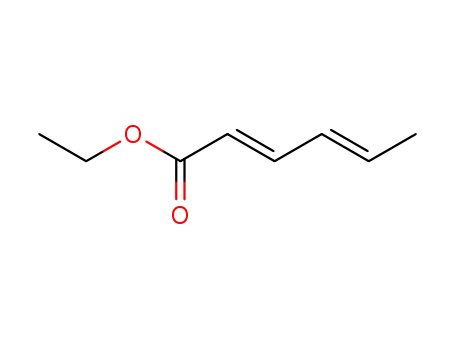
Reaction of (alkoxymethyl)oxiranes with ...
Mechanistic study of the selective hydrogenation of carboxylic acid derivatives over supported rhenium catalysts
Toyao, Takashi,Ting, Kah Wei,Siddiki, S. M. A. Hakim,Touchy, Abeda S.,Onodera, Wataru,Maeno, Zen,Ariga-Miwa, Hiroko,Kanda, Yasuharu,Asakura, Kiyotaka,Shimizu, Ken-ichi
, p. 5413 - 5424 (2019)
The structure and performance of TiO2-su...
Epoxidation of Alkenes with O2 Catalyzed by EuCl3 under Ambient Conditions
Yamanaka, Ichiro,Nakagaki, Katsumi,Akimoto, Takashi,Otsuka, Kiyoshi
, p. 1717 - 1720 (1994)
EuCl3 dissolved in a mixture of propanoi...
LiBH4-promoted Hydroboration of Alkenes with 1,3,2-Benzodioxaborole
Arase, Akira,Nunokawa, Yutaka,Masuda, Yuzuru,Hoshi, Masayuki
, p. 205 - 206 (1991)
In the presence of a small amount of LiB...
Catalytic conversion of ethanol into an advanced biofuel: Unprecedented selectivity for n-butanol
Dowson, George R. M.,Haddow, Mairi F.,Lee, Jason,Wingad, Richard L.,Wass, Duncan F.
, p. 9005 - 9008 (2013)
Taming the beast: Unprecedented selectiv...
Supported nickel-rhenium catalysts for selective hydrogenation of methyl esters to alcohols
Liu, Kaituo,Pritchard, James,Lu, Li,Van Putten, Robbert,Verhoeven,Schmitkamp, Mike,Huang, Xiaoming,Lefort, Laurent,Kiely, Christopher J.,Hensen, Emiel J. M.,Pidko, Evgeny A.
, p. 9761 - 9764 (2017)
The addition of Re to Ni on TiO2 yields ...
Revised Mechanisms of the Catalytic Alcohol Dehydrogenation and Ester Reduction with the Milstein PNN Complex of Ruthenium
Gusev, Dmitry G.
, (2020)
The combined experimental/DFT computatio...
A new route of the reaction of EtAlCl2 with α-olefins catalyzed by Ti complexes
Ibragimov,Khafizova,Zagrebel'naya,Parfenova,Sultanov,Khalilov,Dzkemilev
, p. 292 - 296 (2001)
A new method for the synthesis of dialky...
Catalytic upgrading of ethanol to butanol over a binary catalytic system of FeNiOx and LiOH
Li, Xianquan,Li, Xinsheng,Liu, Shimin,Pang, Jifeng,Wang, Junhu,Wang, Zhinuo,Zhang, Tao,Zheng, Mingyuan
, p. 672 - 678 (2020)
Catalytic conversion of ethanol to butan...
-
Hurd,McNamee
, (1937)
-
Enzymes inhibitory constituents from Buddleja crispa
Ahmad, Ijaz,Malik, Abdul,Afza, Nighat,Anis, Itrat,Fatima, Itrat,Nawaz, Sarfraz Ahmad,Tareen, Rasool Bukhsh,Iqbal Choudhary
, p. 341 - 346 (2005)
Steroidal galactoside 1 and aryl esters ...
-
Fichter,Leupin
, p. 616,622 (1938)
-
Reactivity of active oxygen species generated in the EuCl3 catalytic system for monooxygenation of hydrocarbons
Yamanaka, Ichiro,Nakagaki, Katsumi,Akimoto, Takashi,Otsuka, Kiyoshi
, p. 2511 - 2517 (1996)
The reactivity of active oxygen species ...
In-Situ generation of surface-active HCo(CO)y like intermediate from gold supported on ion-promoted Co3O4 for induced hydroformylation-hydrogenation of alkenes to alcohols
Akinnawo, Christianah A.,Meijboom, Reinout,Mogudi, Batsile M.,Oseghale, Charles O.
, (2020)
In this study, a greener and stable surf...
OXIRANE RINGS: STUDIES AND APPLICATIONS OF A NEW CHEMO AND REGIO SELECTIVE REDUCTIVE OPENING OF EPOXIDES
Bonini, Carlo,Fabio, Romano Di,Sotgiu, Giovanni,Cavagnero, Silvia
, p. 2895 - 2904 (1989)
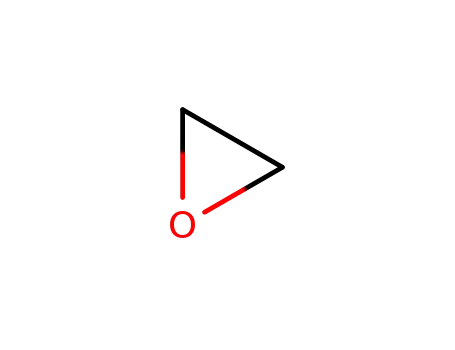
The straightforward reductive opening of...
Lithium Triethylborohydride-promoted Hydroboration of Alkenes with Dialkoxyboranes
Arase, Akira,Nunokawa, Yutaka,Masuda, Yuzuru,Hoshi, Masayuki
, p. 51 - 52 (1992)
In the presence of a catalytic ammount o...
REGIOSELECTIVITY CHANGES IN HEXANE HYDROXYLATION BY IODOZOBENZENE CATALYZED BY TETRAARYLPORPHYRINATOIRON COMPLEXES
Khenkin, Alexander,Koifman, Oskar,Semeikin, Alexander,Shilov, Alexander,Shteinman, Albert
, p. 4247 - 4248 (1985)
Regioselectivity of hexane hydroxylation...
Hydroboration. 54. New General Synthesis of Alkyldihaloboranes via Hydroboration of Alkenes with Dihaloborane-Dimethyl Sulfide Complexes. Unusual Trends in the Reactivities and Directive Effects
Brown, Herbert C.,Ravindran, N.,Kulkarni, Surendra U.
, p. 384 - 389 (1980)
The reactions of alkenes with the dimeth...
PHOTOCHEMICALLY-DRIVEN BIOMIMETIC OXIDATION OF ALKANES AND OLEFINES
Shelnutt, J.A.,Trudell, D.E.
, p. 5231 - 5234 (1989)
Photochemical oxidation of hydrocarbons ...
Alkene-pinacolborane hydroborations catalyzed by lanthanum tris[bis(trimethylsilyl)amide]
Horino, Yoshikazu,Livinghouse, Tom,Stan, Magdalena
, p. 2639 - 2641 (2004)
Tris[bis(trimethylsilyl)amide] has been ...
The Milstein Bipyridyl PNN Pincer Complex of Ruthenium Becomes a Noyori-Type Catalyst under Reducing Conditions
Dawe, Louise N.,Karimzadeh-Younjali, Morteza,Dai, Zengjin,Khaskin, Eugene,Gusev, Dmitry G.
, p. 19510 - 19522 (2020)
Hydrogenation of the dearomatized PNN li...
-
Bigley,Payling
, p. 3974 (1965)
-
Development of a screening system for the evaluation of soybean volatiles
Matsui, Kenji,Kakumyan, Pattana,Kato, Marie,Hajika, Makita
, p. 1844 - 1848 (2009)
Flavor properties are important factors ...
Upgrading ethanol to 1-butanol with a homogeneous air-stable ruthenium catalyst
Tseng, Kuei-Nin T.,Lin, Steve,Kampf, Jeff W.,Szymczak, Nathaniel K.
, p. 2901 - 2904 (2016)
An amide-derived N,N,N-Ru(ii) complex ca...
Homogeneous catalytic hydrogenation of long-chain esters by an osmium pincer complex and its potential application in the direct conversion of triglycerides into fatty alcohols
Acosta-Ramirez, Alberto,Bertoli, Marcello,Gusev, Dmitry G.,Schlaf, Marcel
, p. 1178 - 1188 (2012)
The osmium hydride complexes OsH2(CO)[NH...
-
Cottle,Hollyday
, p. 510,513,514 (1947)
-
-
Zweifel,Arzoumanian
, p. 291,292, 294, 295 (1967)
-
The key role of the latent N-H group in Milstein's catalyst for ester hydrogenation
Chianese, Anthony R.,He, Tianyi,Jarczyk, Cole E.,Keith, Jason M.,Kelly, Sophie. E.,Kim, Thao,Pham, John,Reynolds, Eamon F.
, p. 8477 - 8492 (2021)
We previously demonstrated that Milstein...
Organolanthanide-Catalyzed Hydroboration of Olefins
Harrison, Karl N.,Marks, Tobin J.
, p. 9220 - 9221 (1992)
-
HYDROMAGNESATION OF UNSATURATED COMPOUNDS USING DIETHYLAMINOMAGNESIUM HYDRIDE, CATALYZED BY TRANSITION METAL COMPLEXES
Vostrikova, O. S.,Sultanov, R. M.,Dzhemilev, U. M.
, p. 1724 - 1726 (1983)
-
Biochemical and structural characterisation of a haloalkane dehalogenase from a marine Rhodobacteraceae
Novak, Halina R.,Sayer, Christopher,Isupov, Michail N.,Gotz, Dorothee,Spragg, Andrew Mearns,Littlechild, Jennifer A.
, p. 1616 - 1622 (2014)
A putative haloalkane dehalogenase has b...
Chemoselective hydrogenolysis of tetrahydrofurfuryl alcohol to 1,5-pentanediol
Koso, Shuichi,Furikado, Ippei,Shimao, Akira,Miyazawa, Tomohisa,Kunimori, Kimio,Tomishige, Keiichi
, p. 2035 - 2037 (2009)
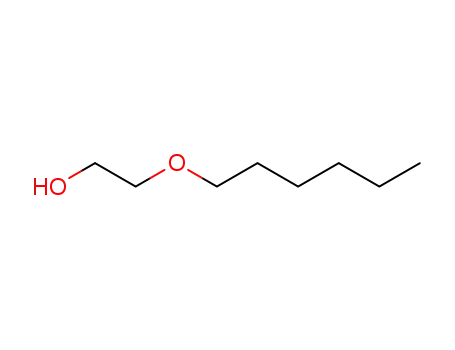
Direct conversion of tetrahydrofurfuryl ...
Continuous flow hydrogenation reactions by Pd catalysts onto hybrid ZrO2/PVA materials
Liguori, Francesca,Barbaro, Pierluigi,Sawa, Haruo
, p. 58 - 65 (2014)
Palladium nanoparticles of 3.2?±?0.9?nm ...
N-methylpyrrolidine-zinc borohydride: As a new stable and efficient reducing agent in organic synthesis
Tajbakhsh,Lakouraj,Mohanazadeh,Ahmadi-Nejhad
, p. 229 - 236 (2003)
N-Methylpyrrolidine-zinc borohydride is ...
One-pot catalytic conversion of cellulose and of woody biomass solids to liquid fuels
Matson, Theodore D.,Barta, Katalin,Iretskii, Alexei V.,Ford, Peter C.
, p. 14090 - 14097 (2011)
Efficient methodologies for converting b...
Selective hydrogenation of 3-Hexyn-1-ol with Pd nanoparticles synthesized via microemulsions
Montsch, Thomas,Heuchel, Moritz,Traa, Yvonne,Klemm, Elias,Stubenrauch, Cosima
, p. 19 - 28 (2017)
In the study at hand we present a design...
Ruthenium pincer-catalyzed acylation of alcohols using esters with liberation of hydrogen under neutral conditions
Gnanaprakasam, Boopathy,Ben-David, Yehoshoa,Milstein, David
, p. 3169 - 3173 (2010)
Acylation of secondary alcohols using no...
Shape Selective Alkane Hydroxylation by Metalloporphyrin Catalysts
Cook, Bruce R.,Reinert, Thomas J.,Suslick, Kenneth S.
, p. 7281 - 7286 (1986)
A series of manganese and iron porphyrin...
Hydrogenation of adipic acid to 1,6-hexanediol by supported bimetallic Ir-Re catalyst
Li, Xiaoyue,Liang, Changhai,Luo, Jingjie
, (2020)
A series of supported Ir-Re catalysts ha...
Upgrading of Ethanol to n-Butanol via a Ruthenium Catalyst in Aqueous Solution
Dibenedetto, Tarah A.,Jones, William D.
, p. 1884 - 1888 (2021)
The upgrading of ethanol to n-butanol vi...
Regiodivergent Reductive Opening of Epoxides by Catalytic Hydrogenation Promoted by a (Cyclopentadienone)iron Complex
De Vries, Johannes G.,Gandini, Tommaso,Gennari, Cesare,Jiao, Haijun,Pignataro, Luca,Stadler, Bernhard M.,Tadiello, Laura,Tin, Sergey
, p. 235 - 246 (2022/01/03)
The reductive opening of epoxides repres...
Hydrogenation of Esters by Manganese Catalysts
Li, Fu,Li, Xiao-Gen,Xiao, Li-Jun,Xie, Jian-Hua,Xu, Yue,Zhou, Qi-Lin
, (2022/01/13)
The hydrogenation of esters catalyzed by...
Chemoenzymatic one-pot reaction from carboxylic acid to nitrile: Via oxime
Hecko, Sebastian,Horvat, Melissa,Klempier, Norbert,Martínková, Ludmila,Pátek, Miroslav,R?disch, Robert,Rudroff, Florian,Schiefer, Astrid,Weilch, Victoria,Wilding, Birgit,Winkler, Margit
, p. 62 - 66 (2022/01/22)
We report a new chemoenzymatic cascade s...
One-Pot Bioelectrocatalytic Conversion of Chemically Inert Hydrocarbons to Imines
Chen, Hui,Tang, Tianhua,Malapit, Christian A.,Lee, Yoo Seok,Prater, Matthew B.,Weliwatte, N. Samali,Minteer, Shelley D.
supporting information, p. 4047 - 4056 (2022/02/10)
Petroleum hydrocarbons are our major ene...