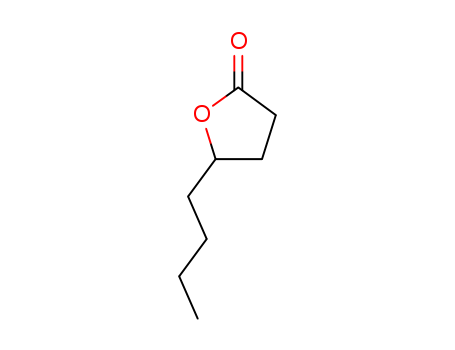
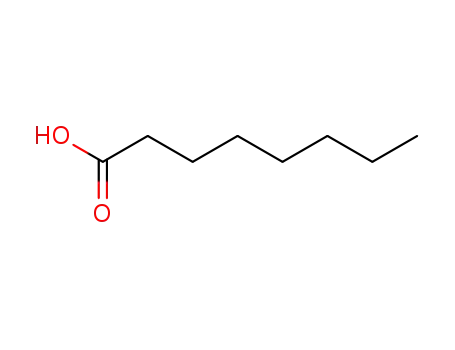
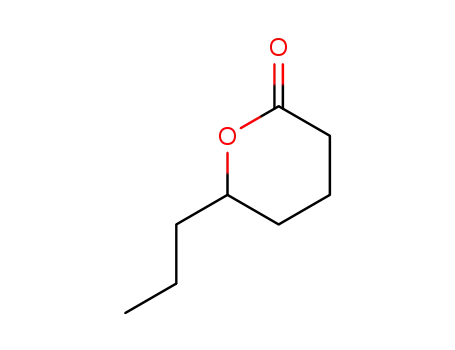
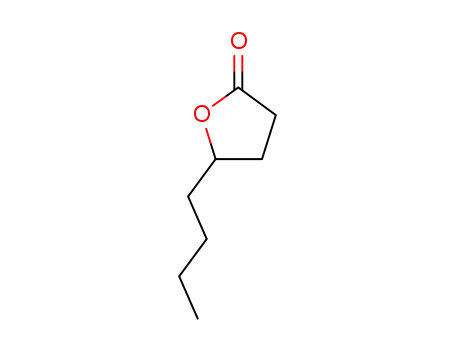
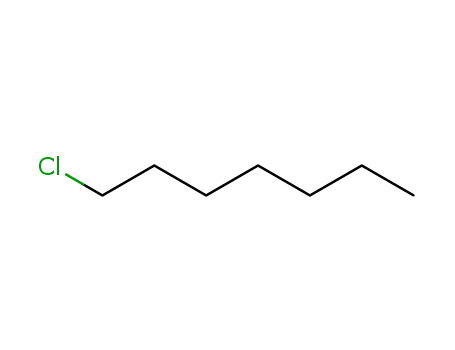
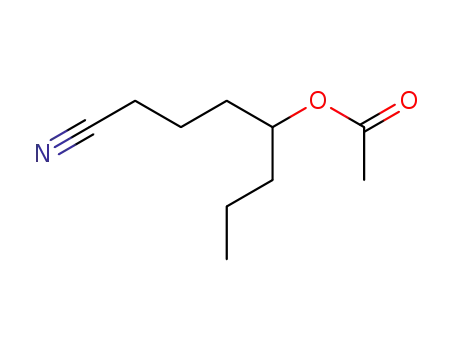
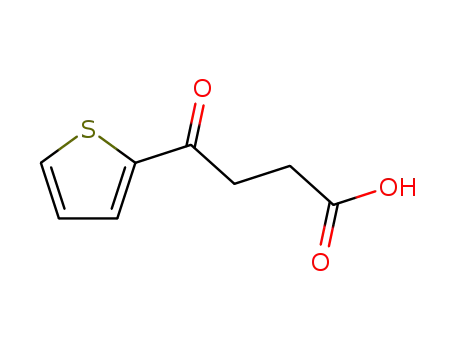
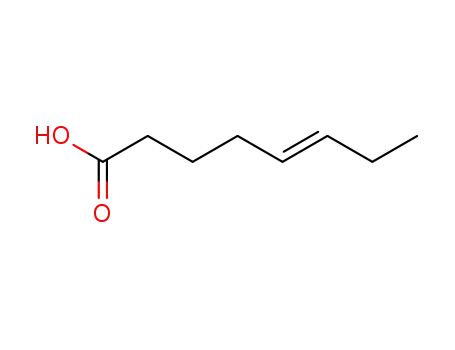
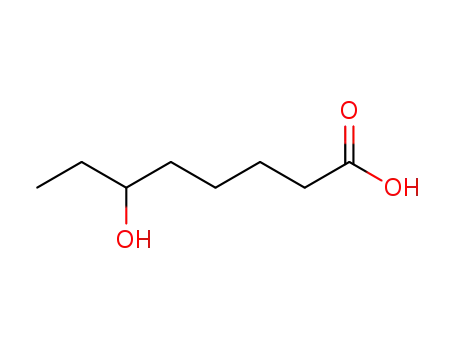
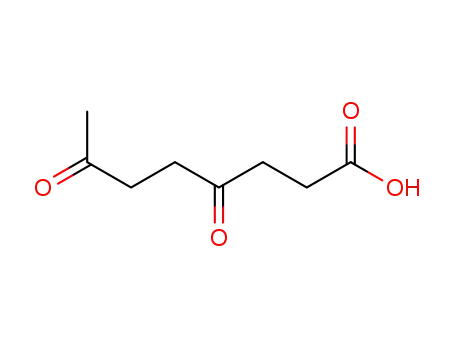
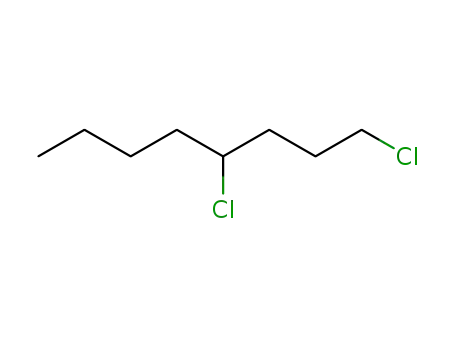
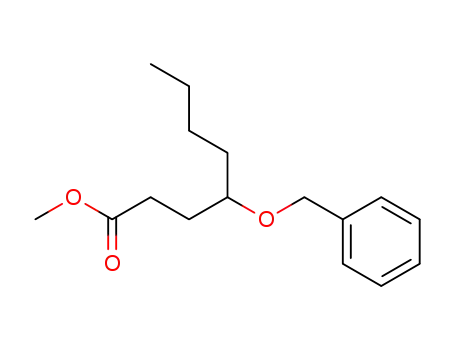
FREE RADICAL ADDITION OF α-BROMOCARBOXYLIC ACIDS TO OLEFINS LEADING TO γ-BUTYROLACTONES
Nakano, Taichi,Kayama, Mikio,Matsumoto, Hideyuki,Nagai, Yoichiro
, p. 415 - 418 (1981)
Benzoyl peroxide-catalyzed free radical ...
DIRECT OXIDATION OF ALKANOIC ACIDS TO LACTONES
Troyanskii, E. I.,Svitan'ko, I. V.,Nikishin, G. I.
, p. 2041 - 2046 (1982)
-
Synthesis of deuterated γ-lactones for use in stable isotope dilution assays
Hislop, J.O.-Anna,Hunt, Martin B.,Fielder, Simon,Rowan, Daryl D.
, p. 7075 - 7083 (2004)
Two syntheses of deuterated γ-lactones f...
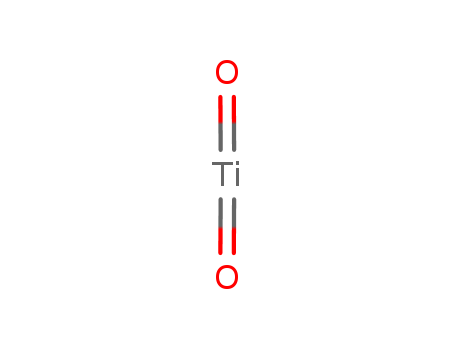
W(OTf)6-Catalyzed Synthesis of Γ-Lactones by Ring Contraction of Macrolides or Ring Closing of Terminal Hydroxyfatty Acids in Ionic Liquid
Xie, Zhong-Yu,Deng, Jin,Fu, Yao
, p. 2332 - 2339 (2018)
γ-Lactones are an important class of fin...
Intermolecular additions of α-boryl radicals
Batey, Robert A.,Pedram, Bijan,Yong, Kelvin,Baquer, Gemma
, p. 6847 - 6850 (1996)
The utility of α-boryl radicals 1 in int...
Free Radical Addition of 2-Bromoalkanoic Acids to Alkenes
Nakano, Taichi,Kayama, Mikio,Nagai, Yoichiro
, p. 1049 - 1052 (1987)
The benzoyl peroxide-catalyzed reaction ...
-
Terai,Tanaka
, p. 822 (1956)
-
A general synthesis of (+)-γ-substituted γ-butyrolactones using a kinetic alkylation-ozonolysis procedure
Gavin,Geraghty
, p. 1351 - 1361 (1994)
A synthesis of (±)-γ-substituted γ-butyr...
-
Okano,M.
, p. 1041 - 1046 (1976)
-
-
Kabasakalian,Townley
, p. 2711,2716 (1962)
-
A mild biosynthesis of lactones via enantioselective hydrolysis of hydroxynitriles
Pollock, Julie A.,Clark, Karen M.,Martynowicz, Bethany J.,Pridgeon, Matthew G.,Rycenga, Matthew J.,Stolle, Kristen E.,Taylor, Stephen K.
, p. 1888 - 1892 (2007)
We have developed a biocatalytic method ...
OXIDATIVE ADDITION OF 1,3-DICARBONYL COMPOUNDS TO OLEFINS BY THE ACTION OF THE Mn(OAc)3/LiCl SYSTEM AND THE SYNTHESIS OF FUNCTIONALLY SUBSTITUTED CYCLOPROPANES
Vinogradov, M. G.,Dolinko, V. I.,Nikishin, G. I.
, p. 334 - 341 (1984)
-
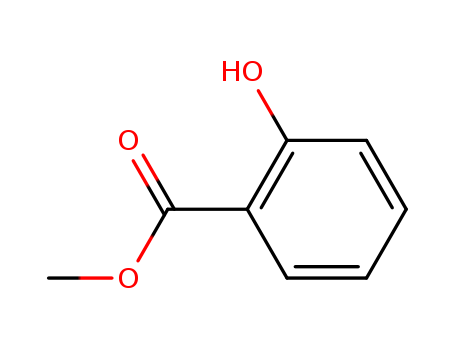
Calcium(II)- And Triflimide-Catalyzed Intramolecular Hydroacyloxylation of Unactivated Alkenes in Hexafluoroisopropanol
Qi, Chenxiao,Yang, Shengwen,Gandon, Vincent,Leb?uf, David
supporting information, p. 7405 - 7409 (2019/10/02)
We report an efficient intramolecular hy...
Photocatalytic Synthesis of γ-Lactones from Alkenes: High-Resolution Mass Spectrometry as a Tool to Study Photoredox Reactions
Triandafillidi, Ierasia,Kokotou, Maroula G.,Kokotos, Christoforos G.
supporting information, p. 36 - 39 (2018/01/17)
A mild photocatalytic manifold for the s...
Electroreductive coupling of aromatic ketones, aldehydes, and aldimines with α,β-unsaturated esters: Synthesis of 5-aryl substituted γ-butyrolactones and lactams
Kise, Naoki,Hamada, Yusuke,Sakurai, Toshihiko
, p. 1143 - 1156 (2017/02/18)
The electroreductive intermolecular coup...