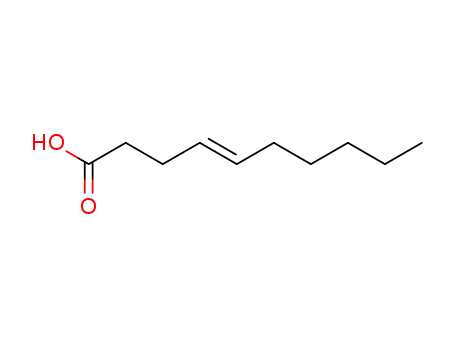Buy Quality Hot Sale GAMMA-DECALACTONE 706-14-9 In Bulk Supply
- Molecular Formula:C10H18O2
- Molecular Weight:170.252
- Appearance/Colour:clear colourless to pale yellow liquid
- Vapor Pressure:0.00852mmHg at 25°C
- Refractive Index:n20/D 1.449
- Boiling Point:266.685 °C at 760 mmHg
- Flash Point:105.788 °C
- PSA:26.30000
- Density:0.947 g/cm3
- LogP:2.66240
- IDLH:1158
- IDLH:3999
gamma-Decalactone(Cas 706-14-9) Usage
|
Chemical Properties
|
γ-Decalactone has a pleasant, fruity, peach-like odor.
|
|
Occurrence
|
Reported found in peach, apricot and strawberry aroma. Also reported in butter, milk, beer, rum, red and white wine, mango, bilberry, plums, prunes, guava, peach, strawberry fruit and cheeses.
|
|
Uses
|
γ-Decalactone may be used as an analytical reference standard for the quantification of the analyte in fruit beverages using different chromatography techniques.
|
|
Preparation
|
Naturalγ-decalactone is produced biotechnologically starting from ricinoleic acid, which is degraded by β-oxidation to 4-hydroxydecanoic acid, which lactonizes at lower pH to yield γ-decalactone
|
|
Aroma threshold values
|
Detection: 1 to 11 ppb
|
|
Taste threshold values
|
Taste characteristics at 10 ppm: creamy, fatty, oily, buttery sweet, coconut, fruity and peach-like.
|
|
Synthesis Reference(s)
|
The Journal of Organic Chemistry, 55, p. 462, 1990 DOI: 10.1021/jo00289a016Synthetic Communications, 18, p. 2241, 1988 DOI: 10.1080/00397918808082366
|
|
General Description
|
γ-Decalactone is a flavor and fragrance compound, which finds applications in food and beverage industry.
|
|
Flammability and Explosibility
|
Notclassified
|
|
Synthesis
|
By heating γ-bromocapric acid in a sodium carbonate solution; by prolonged heating of 9-decen-1-oic acid with 80% H2SO2 at 90°C
|
|
Who Evaluation
|
Evaluation year: 2008
|
InChI:InChI=1/C10H18O2/c1-2-3-4-5-6-9-7-8-10(11)12-9/h9H,2-8H2,1H3/t9-/m0/s1
706-14-9 Relevant articles
Photo-induced radical borylation of hemiacetals via C–C bond cleavage
Liu, Qianyi,Zhang, Jianning,Zhang, Lei,Mo, Fanyang
supporting information, (2021/01/05)
In this study, we reported a photo-induc...
Calcium(II)- And Triflimide-Catalyzed Intramolecular Hydroacyloxylation of Unactivated Alkenes in Hexafluoroisopropanol
Qi, Chenxiao,Yang, Shengwen,Gandon, Vincent,Leb?uf, David
supporting information, p. 7405 - 7409 (2019/10/02)
We report an efficient intramolecular hy...
W(OTf)6-Catalyzed Synthesis of Γ-Lactones by Ring Contraction of Macrolides or Ring Closing of Terminal Hydroxyfatty Acids in Ionic Liquid
Xie, Zhong-Yu,Deng, Jin,Fu, Yao
, p. 2332 - 2339 (2018/07/31)
γ-Lactones are an important class of fin...
Nucleo-Palladation-Triggering Alkene Functionalization: A Route to γ-Lactones
Zheng, Meifang,Chen, Pengquan,Huang, Liangbin,Wu, Wanqing,Jiang, Huanfeng
, p. 5756 - 5759 (2017/11/10)
An unprecedented strategy for the highly...
706-14-9 Process route
-
- 111-66-0,25068-25-1
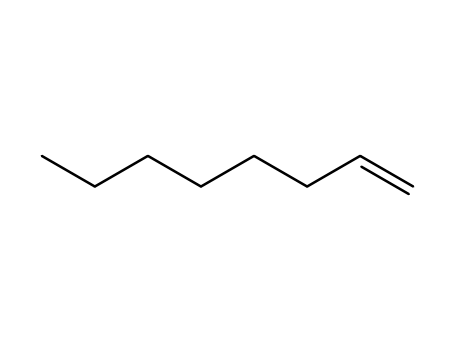
oct-1-ene
-
- 108-24-7
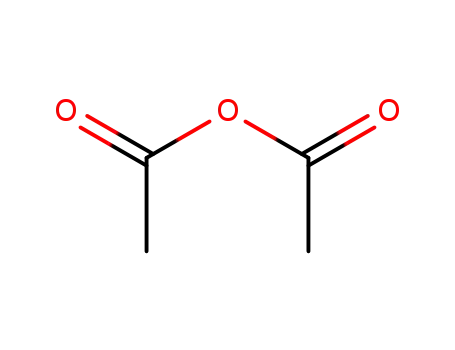
acetic anhydride
-
- 64-19-7,77671-22-8
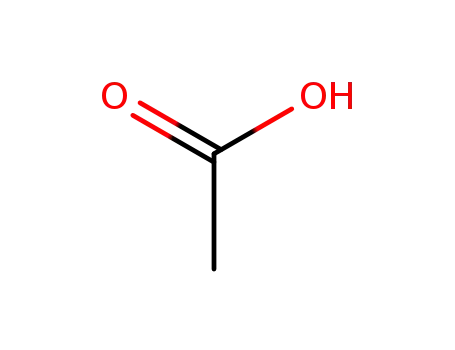
acetic acid
-
- 706-14-9,2825-92-5
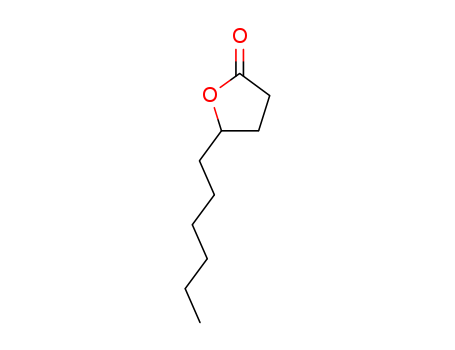
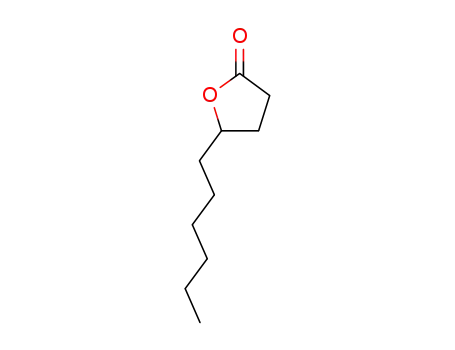
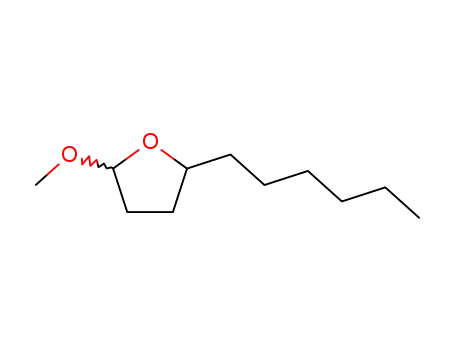
5-hexyldihydro-2(3H)-furanone
-
- 57602-94-5
(E)-4-decenoic acid
-
- 1071156-77-8
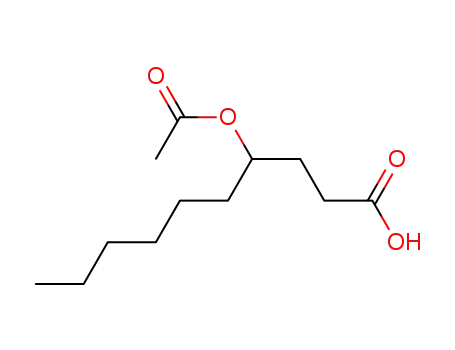
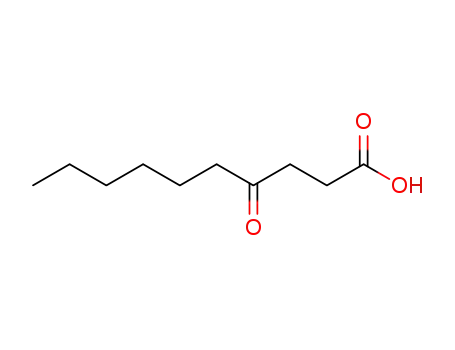
4-Acetoxy-decanoic acid
Conditions
| Conditions |
Yield |
|
With manganese triacetate; at 100 ℃; for 2h; Yield given. Further byproducts given. Title compound not separated from byproducts;
|
4.5 % Chromat.
36 % Chromat.
13 % Chromat. |
|
With manganese triacetate; at 100 ℃; for 2h; Further byproducts given. Title compound not separated from byproducts;
|
6.5 % Chromat.
17 % Chromat.
61 % Chromat. |
-
- 111-66-0,25068-25-1
oct-1-ene
-
- 64-19-7,77671-22-8
acetic acid
-
- 706-14-9,2825-92-5
5-hexyldihydro-2(3H)-furanone
-
- 57602-94-5
(E)-4-decenoic acid
-
- 1071156-77-8
4-Acetoxy-decanoic acid
Conditions
| Conditions |
Yield |
|
With manganese triacetate; at 100 ℃; for 95h; Further byproducts given;
|
0.1 % Chromat.
3.9 % Chromat.
57 % Chromat. |
706-14-9 Upstream products
706-14-9 Downstream products